42 write orbital diagram for mo3+.
Using an orbital box diagram and noble gas notation, show the electron configuration of the gadolinium(III) ion. ... Write out the electron configuration for Mo3+. View Answer.
Apr 1, 2020 · 1 answerMo: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d4. Mo 3+: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s0 4d3. Mo is 42 on the periodic table, since the question asks for ...What is the electronic configuration of nitrogen in the ...2 answersJul 23, 2017What is the electronic configuration of Co3+ and what ...3 answersApr 12, 2018What is the electronic configuration of Ni+2? - Quora3 answersSep 1, 2017Is the electron configuration of Fe+11, found in solar ...4 answersApr 19, 2019More results from www.quora.com
The orbital diagram serves as a representation of all the quantum numbers in an electron configuration. It displays all of the different orientations and ...1 answer · Top answer: The symbol Mo is used to represent a chemical element molybdenum. The atomic number of Mo is 42. Its electron configuration is {eq}\left[...
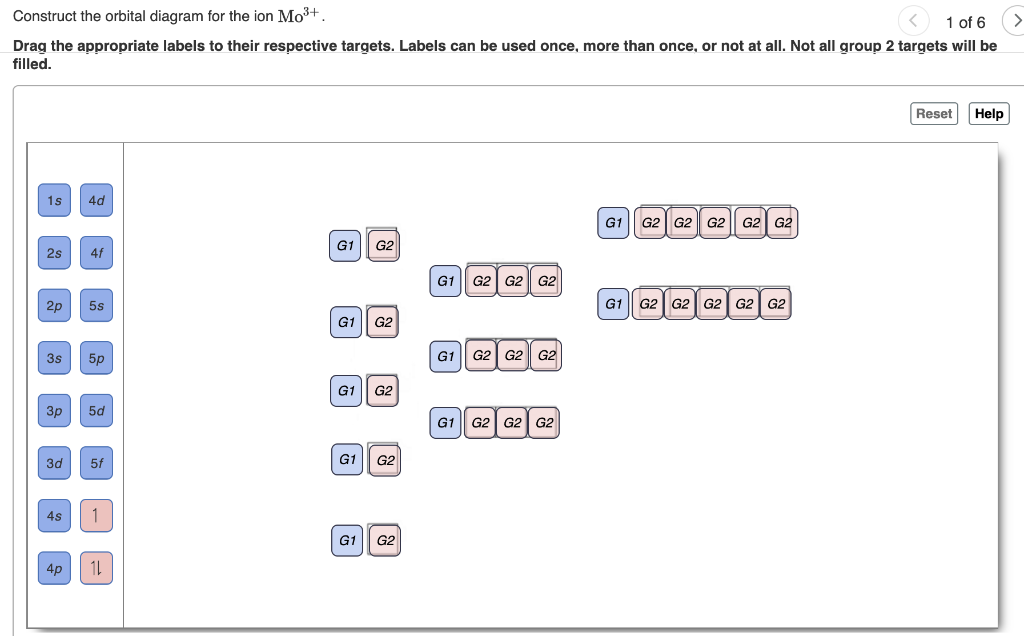
Write orbital diagram for mo3+.
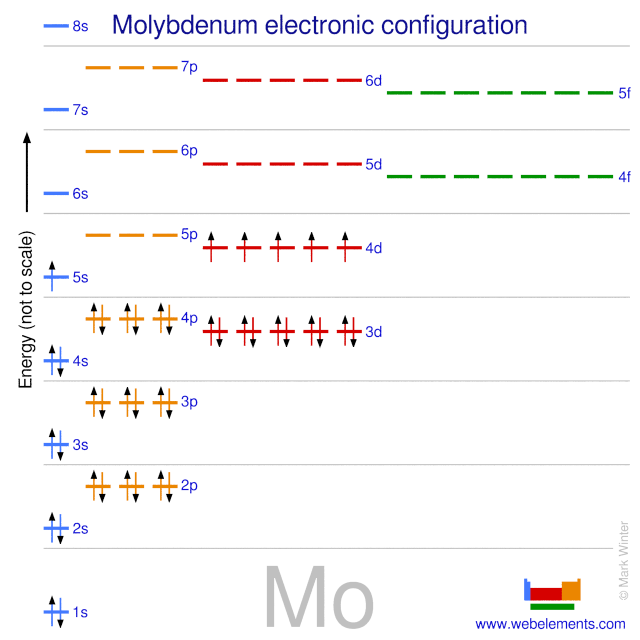
Feb 11, 2016 · 1 answer[Kr]4d3. or. 1s22s22p63s23p63d104s24p64d3. Explanation: The electronic configuration of ground state Mo is. 1s22s22p63s23p63d104s24p64d55s1.
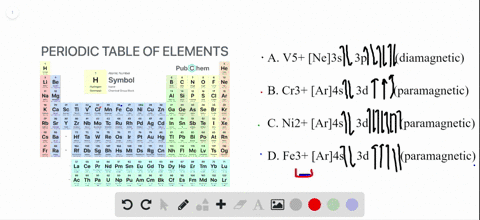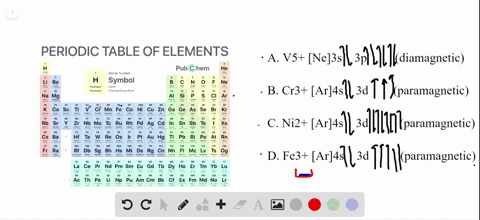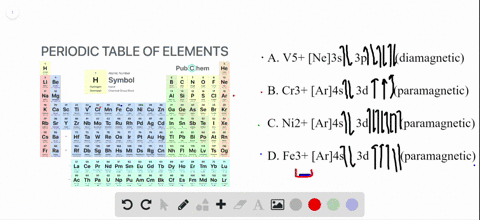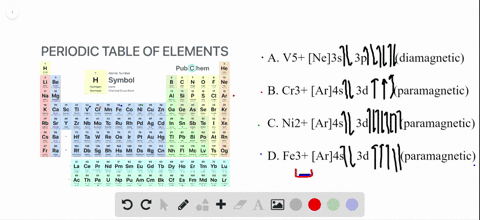
4 answersChapter three problems, seventy eight says to right orbital diagrams for several ions and determined at those ions. Our dia magnetic appear magnetic.
b. Electrons in the 2s orbital are shielded by electrons in the 2p. c. The larger number of electrons found in the 2p orbital leads to greater repulsion. d. The shape of the orbital ultimately determines the energy of the electrons. e. Electrons in the 2s orbital can penetrate the 1s orbital and be closer to the nucleus .
Write orbital diagram for mo3+..
Academia.edu is a platform for academics to share research papers.
Free essays, homework help, flashcards, research papers, book reports, term papers, history, science, politics
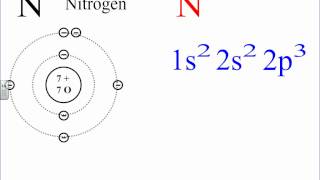
The s or p tell us the orbital block. The superscript tells us the number of electrons in the orbital. The s orbitals Groups 1 & 2 (columns) can hold 2 electrons The p orbitals Groups 13 - 18 (columns) can hold 6 electrons The d orbitals Groups 3-12 (columns) can hold 10 electrons. The f orbitals can hold 14 electrons.
In writing orbital diagrams, first, determine the electron configuration of the neutral atom and remove electrons accordingly.
solucionario quimica de raymond chang 12 edicion . 697 Pages. solucionario quimica de raymond chang 12 edicion
Not to worry! Search thousands of other internships, scholarships and other student programs in 120+ countries. Search Or if you are wondering, who we are:...
Problem: What is the electron configuration of Mo3+? ... We can write out the full configuration, which is one US too to us to two p six, three s two.1 answer · Top answer: [Kr] 4d3
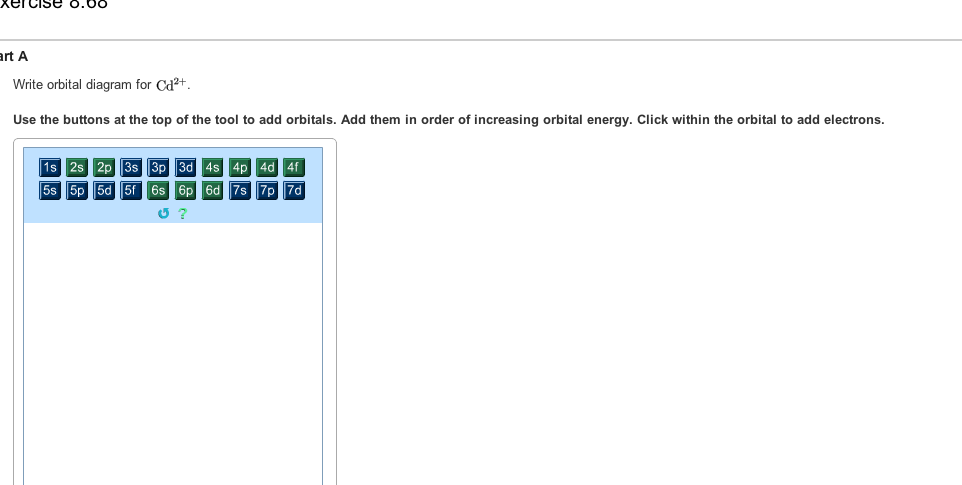
Question: Write orbital diagram for Mo3+ . Use the buttons at the top of the tool to add orbitals. Add them in order of increasing orbital energy.
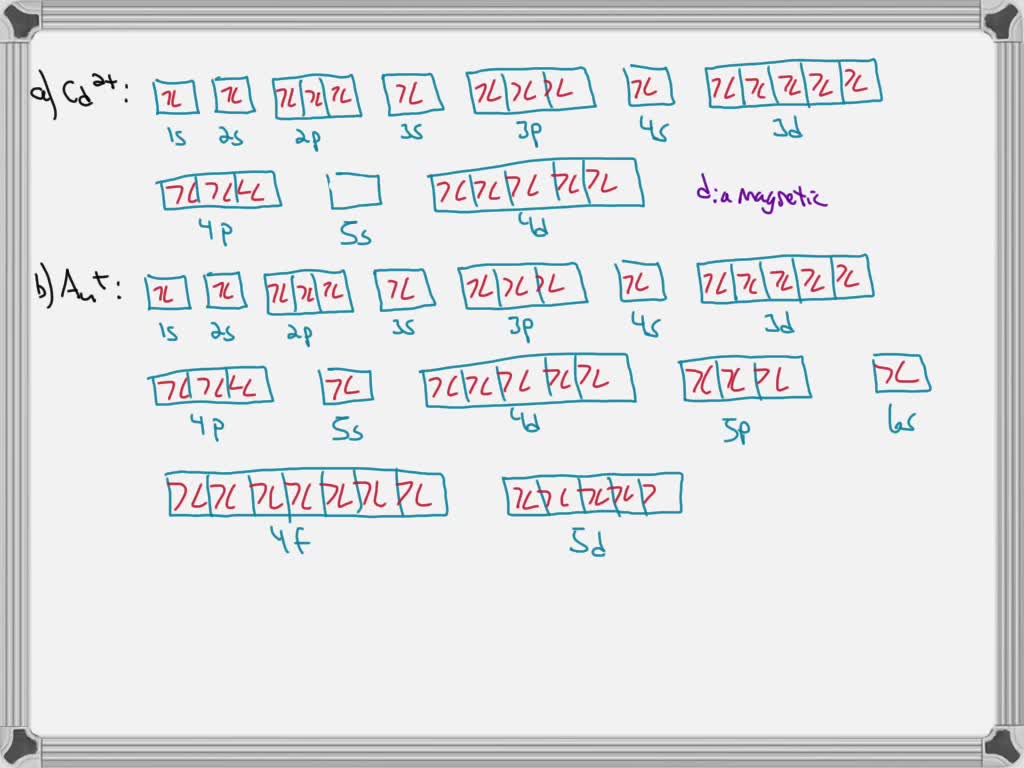
Write orbital diagrams for each ion and determine if the ion is diamagnetic or paramagnetic a cd2 b au c mo3 d zr2 2























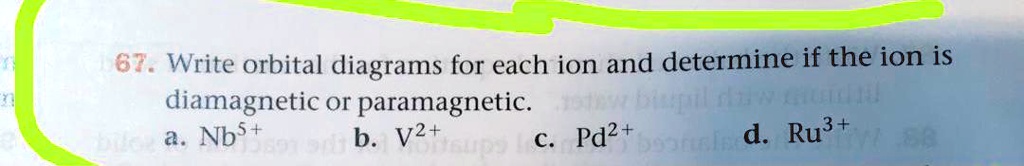




0 Response to "42 write orbital diagram for mo3+."
Post a Comment